Peracetic Acid: An Effective Wastewater Disinfectant
By Jacquelyn N. Wilson

Peracetic acid (PAA) is one of the most versatile and widely used disinfectants on the market. PAA has proven to be an excellent sanitizer and disinfectant in the food, beverage, cosmetic, pharmaceutical, and agriculture industries for industrial water treatment, as well as an excellent disinfectant for use in the wastewater treatment industry.
PAA exists in an equilibrium with hydrogen peroxide, acetic acid, and water, with a potent, non-discriminant, oxidizing chemistry that exhibits higher oxidization potential than common halogen chemistries such as sodium hypochlorite bleach, gaseous chlorine, and chlorine dioxide.
The most common PAA concentrations on the market for wastewater disinfection are 12 percent, 15 percent, and 22 percent active PAA.
PAA is a non-persistent chemistry. Upon addition to water, PAA will degrade naturally over time. In the presence of organic matter, the degradation rate of PAA is increased. PAA constituents eventually break down to their separate components: acetic acid and oxygen. This helps to reduce impacts to the receiving stream or body of water by releasing minimal residual chemicals.
Since PAA is not composed of halogens, such as chlorine, it does not produce harmful disinfection byproducts (DBPs) such as trihalomethanes (THMs). The environmentally friendly nature of PAA is one of the primary reasons for wastewater facility management to seek this alternative.
Wastewater Applications
PAA can be used for the disinfection of all types of wastewater, including primary, secondary, tertiary, and bypass flows, as well as sanitary sewer overflows (SSOs) and combined sewer overflows (CSOs). Dosages of PAA can vary for each of these applications based on background levels of total suspended solids (TSS), color, algae, and reducing agents.
In secondary wastewater facilities, average feed dosages to achieve disinfection range from 1.0 ppm to 3.0 mg/L (ppm) active PAA. The PAA product is typically fed neat. Dilution prior to injection is not recommended because the product will begin to work as soon as it contacts the contaminated water.
PAA feed is best controlled with a flow-paced pump and residual monitoring. Concentration x Time (CT) curves have been developed for better control and efficient use of product. PAA can be fed using diaphragm or peristaltic chemical pumps. It is important to ensure that all equipment is comprised of PAA compatible material (see Figure 1).
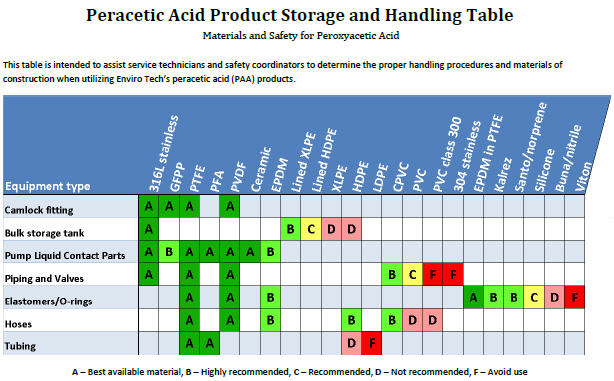
The best materials for construction of feed lines and piping are 316 stainless steel or PTFE/PVDF/HDPE lined steel. PAA residuals can be easily monitored using handheld colorimetric meters using Palin Modified DPD Methodology (Enviro Tech US Patent 7,651,860 B2) or the Palintest Kemio. Online analyzers for measuring PAA residual are available.
Activation
PAA has a quick reaction rate and requires shorter contact times than the industry leading chemical alternatives. In most wastewater systems, recommended contact time to achieve disinfection is 20 to 30 minutes. Disinfection can be achieved in less time if the PAA concentration is increased.
Contact times longer than two hours are not recommended due to organism regrowth potential. If the disinfection contact time is extended beyond an hour, it is recommended to move the PAA injection point farther downstream in the system. This allows for most existing chlorine contact chambers to be easily retrofitted for use with PAA.
Injection sites for PAA are best in areas of turbulence to ensure adequate mixing. Dispersion heads and physical mixers may be used to induce mixing if necessary.
Storage
PAA products should remain in original containers or designated bulk tanks. Use of containers not approved for PAA use can cause contamination and unsafe conditions. PAA products for wastewater applications are readily available in containers ranging from five-gallon pails to 330-gallon totes and 4000-gallon bulk loads. The product should be stored at ambient temperature out of direct sunlight, though freezing points are for all formulations are around -14°F. In these conditions, the shelf-life of PAA is at least one year.
Bulk storage tanks should be made of passivated 316L stainless steel construction for best results. Alternative options that will still provide suitable longevity are also available. All tanks require a pressure relief system or 4” vent and a total or low-point drain. Chemical off-loads are typically handled by PAA suppliers, limiting operations staff exposure to hazardous conditions.
Safety And Handling
PAA should always be handled with care. The recommended personal protective equipment (PPE) when the product in its neat form is goggles, face shield, chemical-resistant apron, and chemical-resistant gloves. When PAA is diluted, vented safety glasses and latex or vinyl gloves are recommended.
It is important to avoid contamination of PAA; most product decomposition or loss in concentration strength are due to contamination issues. To prevent product contamination, do not use unauthorized containers for PAA storage, add PAA to water only, and never return PAA to original containers after removal. In totes and drums, adapters are available to limit PAA contamination.
Transitioning To PAA Disinfection
Here are some recommendations for beginning a transition to PAA-based wastewater disinfection:
- Work collaboratively. Expert firms and consultants can guide pilot programs, develop system design, and support end users in PAA application
- Determine PAA will work effectively in your system by performing background water chemistry analysis to rule out competing materials.
- Test efficacy of PAA products. Measure and achieve permitting-level compliance for fecal coliform and other effluent pathogens during both normal operation and wet-weather periods.
- Perform full-scale trial to evaluate cost savings and ROI. Data collected may include feed rates, number of thousands of gallons treated, and chemical usage, as well as other metrics. Adjust chemical usage accordingly for maximum effectiveness and ROI.
For More Information
- Peracetic Acid Wastewater Disinfection
- Peracetic acid: Structural elucidation for applications in wastewater treatment.
- Peracetic Acid Full-Scale Trial for Disinfection of Secondary Effluent
Jacquelyn N. Wilson is the technical sales manager for Enviro Tech Chemical Services. Previously she worked as a consultant, focusing on the development and use of peracetic acid as an alternative disinfectant in municipal wastewater. She holds a Bachelor of Science degree from Eastern Michigan University.
