Mixed-Mode Chromatography for mAb S Aggregate Removal: Comparison Of Various Mixed-Mode Media To Determine The Optimal Choice For Efficient Purification Process
By Xuemei He
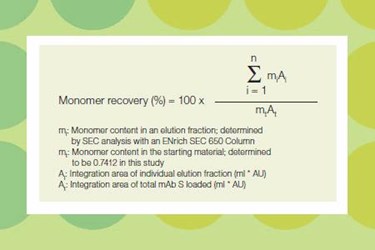
access the Application Note!
Log In
Get unlimited access to:
Trend and Thought Leadership Articles

Case Studies & White Papers

Extensive Product Database

Members-Only Premium Content

Welcome Back! Please Log In to Continue.
X
Enter your credentials below to log in. Not yet a member of Water Online? Subscribe today.
Subscribe to Water Online
X
Subscribe to Water Online
