Innovations In UV Oxidation Direct Conductivity TOC Measurement To Improve Accuracy And Precision
By Roger Schmid and Randy Turner
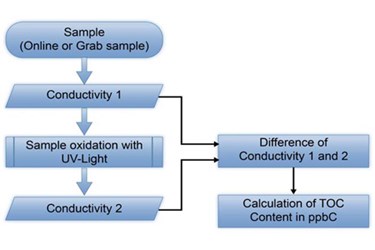
In the pharmaceutical industry the quality guidelines for the production of applied water are set at a particularly high level. In order to guarantee the quality of the ultra‐pure water at all times, more and more pharmaceutical producers employ an automatic and continuous monitoring of processing plants and of ultra‐pure water cycles. The introduction of the TOC content as a specification for ultra‐pure water in the US pharmacopeia in 1998 was an important step for this development. Similar to the standards of the inorganic load of the water (conductivity), the organic load can be verified by a sum parameter. With these changes by the controlling authorities, the pharmaceutical industries can now monitor the organic load of ultra‐pure water continuously and promptly. The manufacturers of monitoring instruments already have experience with this parameter from other pure and ultra‐pure water producing industries (e.g., semiconductor industries).
The manufacturers have developed various techniques to oxidize the organic carbon and the resulting carbon dioxide can be measured. SWAN Analytical Instruments employs the direct UV oxidation method.
This report gives a brief outline of the general measuring methods. Further, it illustrates the possibilities and limitations of the direct UV oxidation, as well as which technical modifications were made to the monitoring instrument, AMI Line TOC, to improve the performance of the UV reactor and, therefore, the precision of the instrument.
Get unlimited access to:
Enter your credentials below to log in. Not yet a member of Water Online? Subscribe today.
