From Compliance To Circularity: Rethinking Chromium Management In Chrome Tanning
By Miguel Ochoa

The Problem: Valuable Chromium Becomes Hazardous Sludge
Chrome tanning represents approximately 80–90% of global leather production due to its performance advantages in hydrothermal stability and mechanical strength.1 Yet fixation efficiency typically ranges between 60% and 80%, meaning a significant fraction of chromium remains in spent tanning liquors.2
The conventional solution is alkaline precipitation, converting dissolved chromium into chromium hydroxide sludge. While effective for regulatory compliance, this approach transforms a reusable raw material into a disposal burden — often requiring hazardous waste management depending on leachability criteria.5 A valuable reagent becomes regulated waste.
Evolving Regulatory And Environmental Risk
Historically, chromium(III) has been regarded as significantly less hazardous than chromium(VI), and chrome tanning systems are designed around its trivalent chemistry. However, modern environmental management increasingly evaluates long-term risk rather than immediate speciation alone.
Under alkaline landfill conditions and in the presence of oxidizing minerals such as manganese oxides, Cr(III) may slowly oxidize to Cr(VI). While this transformation is not inherent to tanning operations, it represents a potential risk pathway once chromium is concentrated in sludge and transferred to landfills or storage environments.
Regulatory frameworks in both North America and Europe have progressively tightened oversight of heavy metals in industrial residues. Chromium-containing wastes may require hazardous classification depending on leachability testing. Chemical management policies increasingly emphasize lifecycle risk and traceability of metal-bearing materials.
At the same time, disposal costs for metal-containing sludges continue to rise, and environmental liability increasingly extends beyond initial discharge compliance. In this context, precipitation does not eliminate chromium — it simply transfers it from the liquid phase to a solid waste stream.
The strategic question is no longer how to meet discharge limits, but how to prevent valuable chromium from becoming an environmental and economic liability.
In light of these evolving regulatory and economic pressures, technologies that prevent chromium from entering the waste stream — rather than stabilizing it as sludge — merit renewed evaluation. The objective therefore shifts from treatment compliance to material stewardship. Recovery-based systems represent not a novel concept, but a strategically aligned response to modern environmental management expectations.
Early investigations in the mid-1990s, conducted under the academic supervision of Professors Judith de Fuentes and Pablo Baricelli, and developed experimentally by Miguel Ochoa and Zayda Esteves demonstrated that chromium(III) from tanning liquors can be adsorbed onto silica-based media under laboratory conditions. However, these investigations focused on demonstrating adsorption feasibility at the laboratory scale rather than developing integrated industrial recovery systems. Since then, research has explored various adsorbent technologies for chromium removal and recovery. The present article revisits regenerable adsorption within a practical, modular framework designed for industrial implementation and regulatory validation.
The goal is not removal; the goal is recovery.
Where Recovery Makes Sense
Chromium losses occur primarily in:
- Spent tanning baths
- Initial rinse waters
- Mixed effluent streams if not segregated
The highest recovery potential exists immediately after drum discharge — before dilution or precipitation. Interception at this point preserves concentration and simplifies recovery.
In a medium-size tannery processing 20 tons of hides per day, chromium losses can exceed several hundred kilograms per week depending on fixation efficiency and bath management practices. For such facilities, even modest chromium recovery efficiencies can translate into significant reductions in both chemical purchasing and hazardous sludge disposal costs.
A Modular Recovery Model
Rather than prescribing a rigid system, the proposed framework consists of three adaptable stages.
1. Chrome Stream Segregation
A dedicated chrome liquor line prevents dilution with beamhouse wastewater. Many facilities already implement this when operating precipitation systems. Segregation is the foundation of recovery.
2. Pretreatment: Protecting the Adsorption Column
Pretreatment removes suspended solids and fats while maintaining chromium in the dissolved phase.
Baseline Physical Pretreatment:
- Screening
- Equalization or settling
- Filtration adapted to plant conditions
Enhanced Clarification (when required):
- Additional clarification under acidic conditions
- No upstream pH increase
- Recycle of filtrates to preserve dissolved chromium
Unlike precipitation, this system avoids intentionally converting chromium into a solid phase. Verification should include:
- Chromium mass balance
- Solids testing
- Leachability confirmation where applicable
The objective is to keep chromium in solution until selective recovery.
3. Adsorption and Chromium Recovery
The pretreated chrome liquor is directed to a packed adsorption column containing pelletized silica gel. Under acidic conditions typical of tanning liquors, chromium(III) ions interact with hydroxyl functional groups on the silica surface and are retained within the bed while the treated liquor exits the column.
When the adsorption bed approaches breakthrough, the column is taken offline and regenerated using dilute hydrochloric acid. The acid desorbs the retained chromium, producing a concentrated chromium chloride (CrCl₃) solution.
This recovered chromium stream can be further processed to produce basic chromium sulfate (BCS), allowing the chromium to be reintroduced into tanning operations. In this way, the system closes the material loop, transforming what would otherwise become waste sludge into a reusable process input.
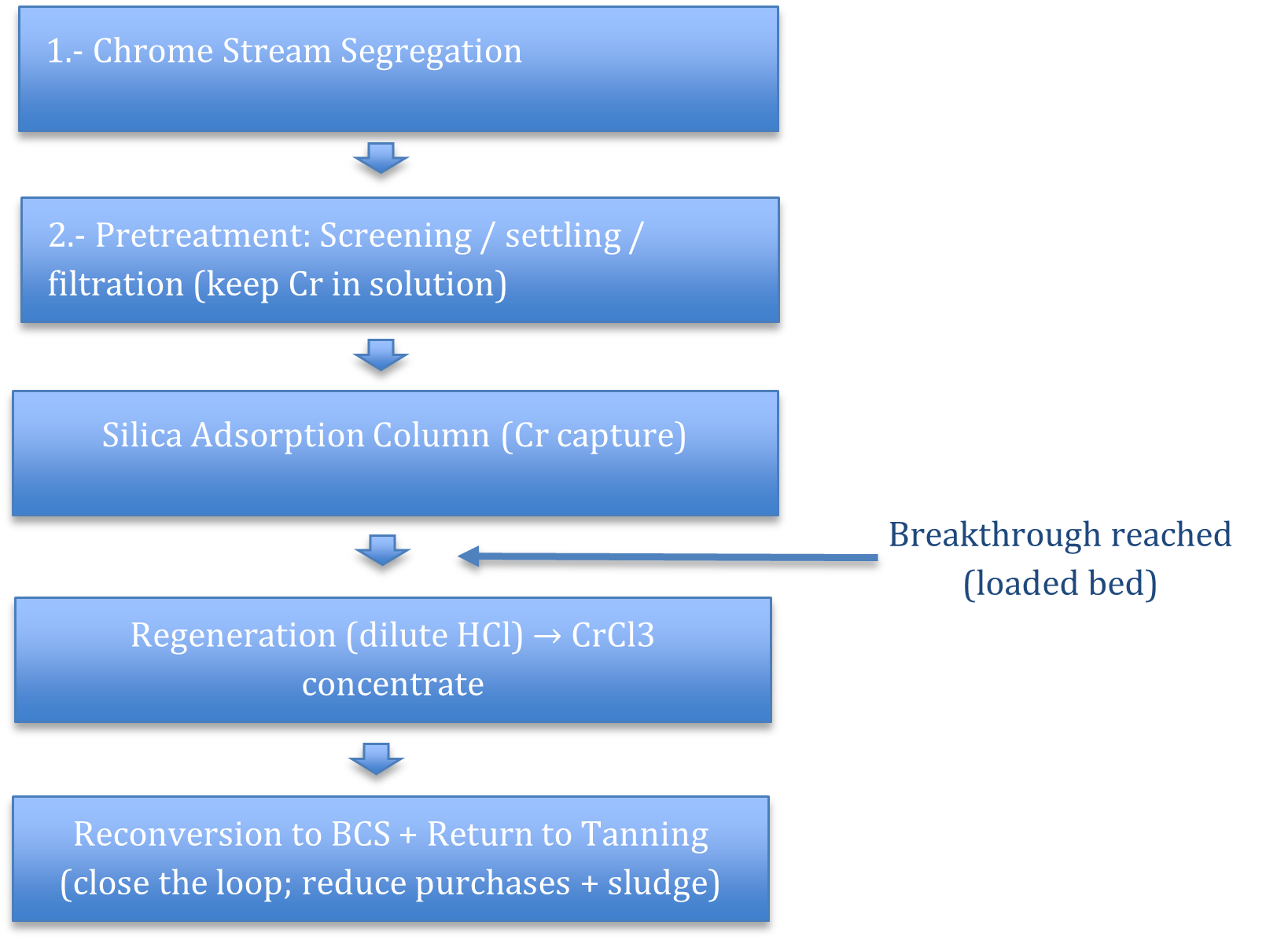
Figure 1. Modular Chromium Recovery Model for Chrome Tanning
Why Pelletized Silica Gel?
Pelletized silica gel provides:
- Stability in acidic chrome liquor
- Mechanical properties in packed beds
- Regeneration with diluted acid
- Industrial availability at reasonable cost
Silica surfaces contain hydroxyl functional groups capable of reversible interaction with Cr³⁺ ions. Adsorption behavior can be described using established isotherm models such as Langmuir-type systems.4
Alternative regenerable media, such as chelating ion exchange resins, may be evaluated depending on site-specific economics and footprint constraints. The recovery architecture itself is not dependent on a single material.
Column Operation: Downflow Or Upflow?
The adsorption unit is typically implemented as a vertically packed column. Operation may be configured as:
- Downflow (conventional and mechanically simple)
- Upflow (advantageous when residual solids are present)
Upflow can reduce surface mat formation and facilitate gas disengagement. Hydraulic loading must be controlled to prevent bed expansion. Flow direction should be selected based on feed characteristics and operational preference rather than imposed as a fixed requirement.
Regeneration And Reuse
When breakthrough is reached:
- The bed is rinsed
- Dilute hydrochloric acid is applied
- A concentrated CrCl₃ stream is recovered
This concentrate can be reconverted into basic chromium sulfate (BCS) and returned to tanning operations.1
Instead of purchasing entirely new chromium salt, the facility reintroduces recovered chromium back into production. The loop is closed.
Recovery vs. Precipitation
The operational implications of both approaches can be summarized as follows:

Precipitation removes chromium, regenerable adsorption recovers it. Unlike precipitation sludge, which intentionally concentrates chromium into a hydroxide solid phase, the proposed system maintains chromium in solution until controlled regeneration and reuse. Hazardous waste classification, where applicable, should be verified using established leachability criteria.5
Validation Framework
Because every tannery operates differently, site-specific validation is essential. A pilot program should evaluate:
- Chromium mass balance
- Breakthrough behavior
- Regeneration efficiency
- Pressure drops
- Leachability of pretreatment solids
Pilot validation not only confirms performance but also provides the documentation required for regulatory acceptance and internal capital approval. Measured performance — not assumptions — determines feasibility.
Conclusion
Regenerable adsorption using pelletized silica gel offers a practical pathway to shift chromium management from disposal to recovery.
By maintaining chromium in solution until controlled regeneration, facilities can:
- Reduce hazardous sludge generation
- Recover valuable metal
- Decrease long-term disposal dependency
- Mitigate potential lifecycle regulatory risk
- Align with circular manufacturing principles
Precipitation was designed for compliance. Recovery is designed for sustainability and resource efficiency.
In an industrial landscape increasingly shaped by lifecycle accountability, material traceability, and rising disposal costs, chromium recovery is not merely a technical alternative — it is a strategic upgrade, where wastewater can become a resource stream.
References
- Covington, A.D. (2009). Tanning Chemistry: The Science of Leather. Royal Society of Chemistry.
- Buljan, J., Reich, G., & Ludvik, J. (2000). Mass Balance in Leather Processing. UNIDO.
- Sreeram, K.J., & Ramasami, T. (2003). Sustaining tanning process through conservation, recovery and better utilization of chromium. Resources, Conservation and Recycling, 38(3), 185–212.
- Foo, K.Y., & Hameed, B.H. (2010). Insights into the modeling of adsorption isotherm systems. Chemical Engineering Journal, 156, 2–10.
- U.S. Environmental Protection Agency (2023). 40 CFR §261.24 – Toxicity Characteristic.
Miguel Ochoa is a Chemical Engineer with over 18 years of experience in industrial manufacturing, process optimization, and environmental systems. His work integrates practical plant operations with resource efficiency, automation, and sustainable process design.
Based in South Florida, he leads Ochoa Parts Automation, supporting industrial clients in water and wastewater treatment instrumentation, process control, and performance improvement initiatives.
